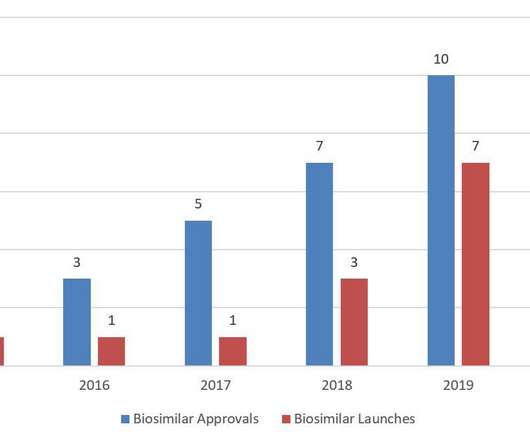
Biosimilars 2020 Year in Review
Fish & Richardson Trademark & Copyright Thoughts
FEBRUARY 4, 2021
Pfizer/Hospira). Pfizer/Hospira). As of the March 23, 2020 transition, however, all “biological product” applications previously approved under the FD&C Act were “deemed” approved under the PHS Act, and companies can now seek biosimilars and interchangeables of these biological products. January 3, 2019. adalimumab-adaz.











Let's personalize your content