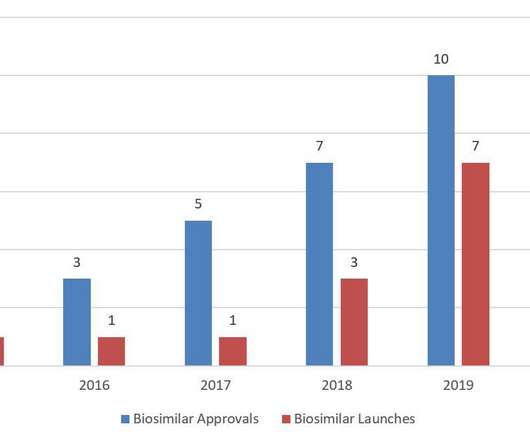
Biosimilars 2020 Year in Review
Fish & Richardson Trademark & Copyright Thoughts
FEBRUARY 4, 2021
In particular, under the new amendments, reference product sponsors (RPSs) are required to provide FDA with copies of any patent lists, along with patent expiration dates, within 30 days of when they were first provided to biosimilar applicants as part of the patent dance (pursuant to 42 U.S.C. § 262( l )(3)(A) or ( l )(7)). and Immunex. .







Let's personalize your content