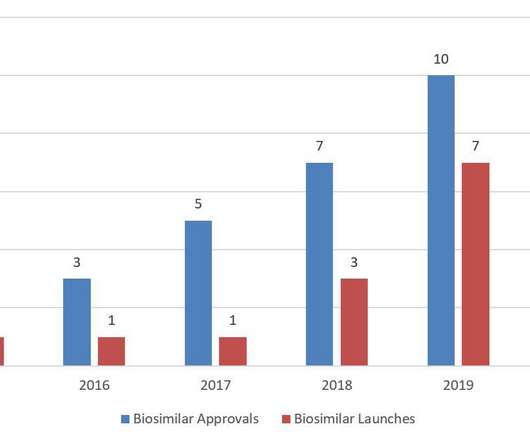
Biosimilars 2020 Year in Review
Fish & Richardson Trademark & Copyright Thoughts
FEBRUARY 4, 2021
BI argues that FDA’s current interpretation “encourages, or at least permits, brand sponsors to use minor concentration changes as an anti-competitive tactic.” license and the ability to bring a biosimilar to market in March 2025. BI points to Humira ® (adalimumab) as an example. A jury trial is scheduled for May 17, 2021.







Let's personalize your content