Fish Attorneys Author Biosimilar Development Article, “Regulatory And Legislative Actions Set The Tone For Biosimilars In 2021”
Fish & Richardson Trademark & Copyright Thoughts
MARCH 8, 2021
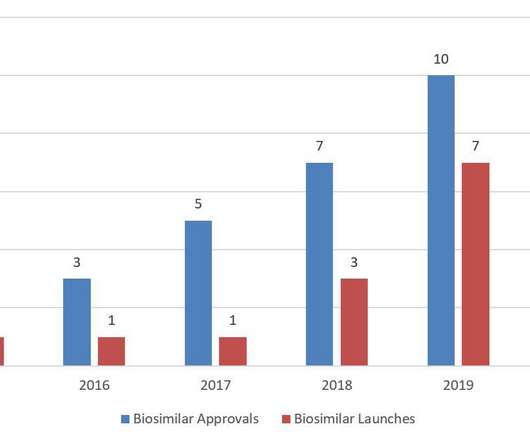
In particular, the authors review FDA’s preparation for the March 23, 2020 transition date, as well as FDA’s efforts to address biosimilar competition, interchangeability, and the COVID-19 pandemic. As of FY 2020 Q4, there were 84 biosimilar development programs enrolled in the biosimilar biological product development (BPD) program.









Let's personalize your content