Biosimilars 2020 Year in Review
Fish & Richardson Trademark & Copyright Thoughts
FEBRUARY 4, 2021
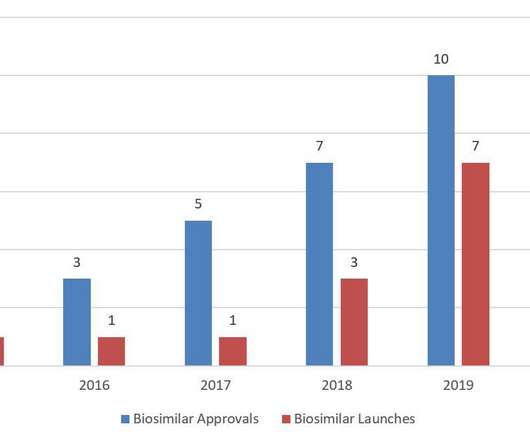
For the first time since FDA licensed the first biosimilar, Sandoz’s Zarxio ® (filgrastim-sndz), in 2015, the United States saw a decrease in annual biosimilar approvals in 2020. BI argues that FDA’s current interpretation “encourages, or at least permits, brand sponsors to use minor concentration changes as an anti-competitive tactic.”









Let's personalize your content