Biosimilars 2020 Year in Review
Fish & Richardson Trademark & Copyright Thoughts
FEBRUARY 4, 2021
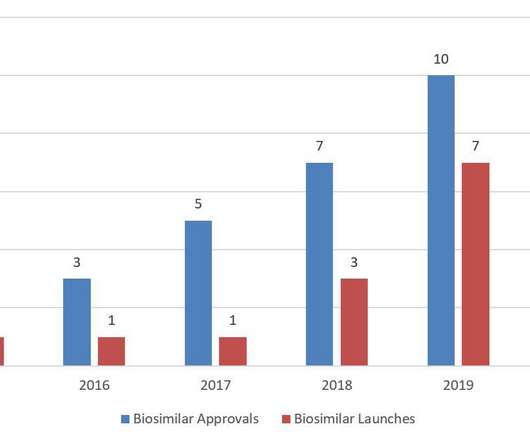
The biosimilar pathway was designed to increase competition for biologics and reduce healthcare costs. For the first time since FDA licensed the first biosimilar, Sandoz’s Zarxio ® (filgrastim-sndz), in 2015, the United States saw a decrease in annual biosimilar approvals in 2020. 2015; resubmitted Feb. 2015; resubmitted Dec.









Let's personalize your content