Biosimilars 2020 Year in Review
Fish & Richardson Trademark & Copyright Thoughts
FEBRUARY 4, 2021
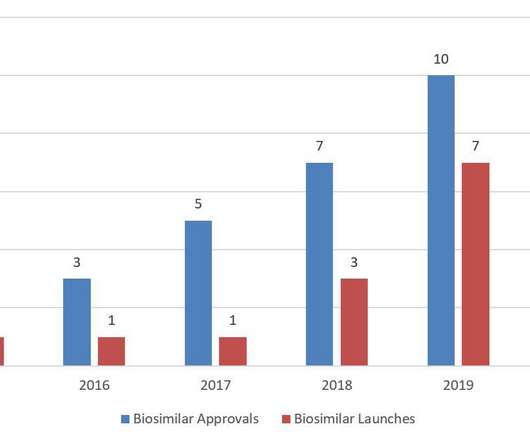
As of March 23, 2020, Mylan’s NDA for Semglee™ was “deemed” to be a BLA, and Mylan has announced its submission of “all necessary documentation” to FDA for approval of Semglee™ as a biosimilar and interchangeable biosimilar product. Market uptake of biosimilars has generally trended upwards in 2020. Not yet launched. etanercept).









Let's personalize your content