Biosimilars 2020 Year in Review
Fish & Richardson Trademark & Copyright Thoughts
FEBRUARY 4, 2021
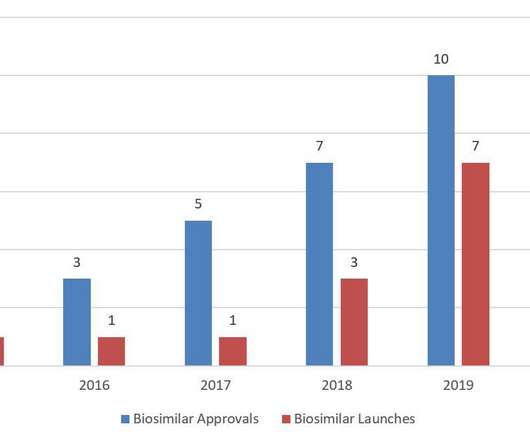
The biosimilar pathway was designed to increase competition for biologics and reduce healthcare costs. Note that FDA still has not designated any biosimilar as interchangeable to its reference product. In 2020, FDA continued to focus on biosimilars through guidance documents, citizen petitions, and other mechanisms.









Let's personalize your content