Biosimilars 2020 Year in Review
Fish & Richardson Trademark & Copyright Thoughts
FEBRUARY 4, 2021
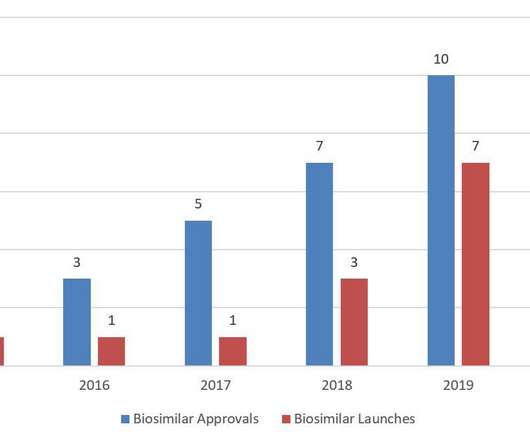
For the first time since FDA licensed the first biosimilar, Sandoz’s Zarxio ® (filgrastim-sndz), in 2015, the United States saw a decrease in annual biosimilar approvals in 2020. In 2020, FDA continued to focus on biosimilars through guidance documents, citizen petitions, and other mechanisms. BPCIA Litigation. Antitrust Litigation.









Let's personalize your content