Brand-Generic Drugs: Infringement Judged by the ANDA Filing
Patently-O
AUGUST 19, 2022
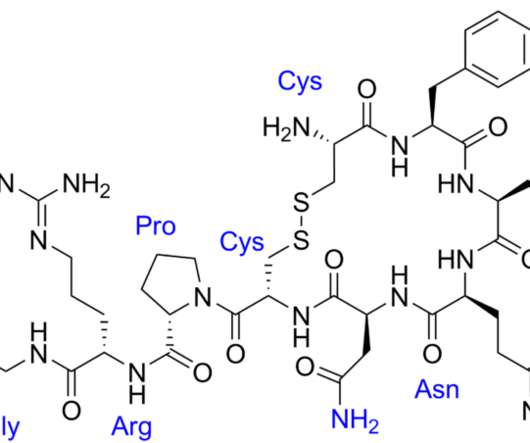
In 2018, Eagle filed its Abbreviated New Drug Application (ANDA) with the FDA seeking to market a generic version of Par’s vasopressin injection product (Vasostrict) used for emergency blood pressure treatment. We have the ANDA filing, but there are always substantial design choices before final release. Eagle Pharm.,



































Let's personalize your content