Biologics and Biosimilars Landscape: IP, Policy, and Market Developments
JD Supra Law
APRIL 8, 2024
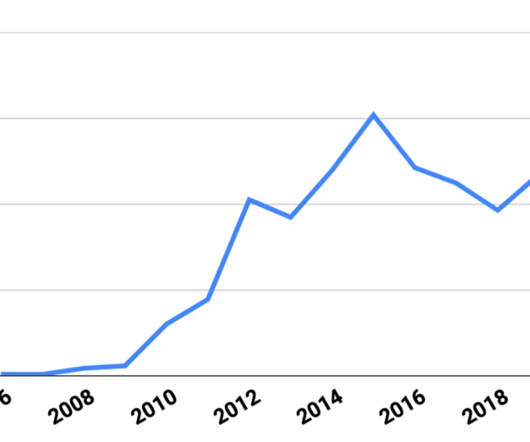
In this review, we discuss the most important recent developments in the biosimilars space, including new biosimilar approvals and launches, litigation under the Biologics Price Competition and Innovation Act (BPCIA), post-grant disputes on biologic drug patents before the United States Patent and Trademark Office (USPTO), and proposed legislation (..)
















































Let's personalize your content